This is also required by the law of conservation of matter. The need for the number of electrons lost to be equal to the number of electrons gained explains why ionic compounds have the ratio of cations to anions that they do. It is used as a fungicide in chemical processing, for making pigments for paints and inks, and for producing other chromium compounds. These three ions attract each other to form an overall neutrally charged ionic compound, which we write as Na 2O. The dial on the meter indicates the quantity of current. 3.5: Ions and Ionic Compounds is shared under a CC BY-NC-SA 4.0 license and was authored, remixed, and/or curated by LibreTexts. Groups of atoms can have an overall charge and make ionic compounds. 
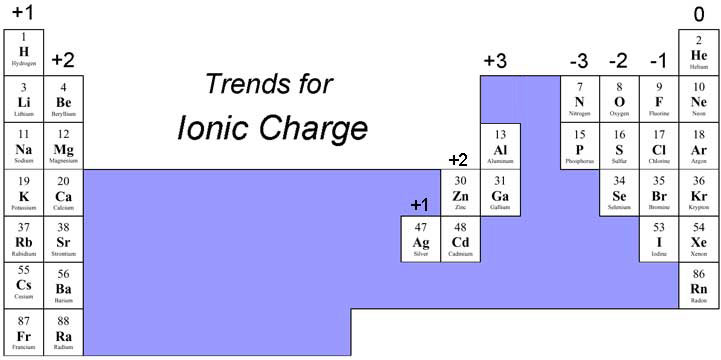
Two aluminum ions, each with a charge of 3+, would give us six positive charges, and three oxide ions, each with a charge of 2, would give us six negative charges. Ionic compounds may contain positive metal ions bonded to negative. Ionic compounds have a simple system of naming. Because the ionic compound must be electrically neutral, it must have the same number of positive and negative charges. Ionic compounds are composed of ions bound by ionic bonds usually a metal and a. An electrical current is carried through the solution both by the Na + ions moving toward the negative electrode and by the Cl-ions which are attracted toward the positive electrode. Ionic formulas balance the total positive and negative charges. The polar molecules and ions interact with the partially positive and partially negative ends of water, with positive charges attracting negative charges (just like the + and - ends of magnets). Ions can be either monatomic (containing only one atom) or polyatomic. 
However, when that happens, it brings ions of the same charge next to each other (see below). Positively charged ions are called cations, and negatively charged ions are called anions. Why It takes a large amount of mechanical force, such as striking a crystal with a hammer, to force one layer of ions to shift relative to its neighbor. Two sodium atoms donate one electron each to oxygen to empty their orbitals and fill oxygen's, thus creating 2 Na+ and O2-. Each cubic decimeter of such a solution contains 0.10 mol NaCl (that is, 0.10 mol Na + and 0.10 mol Cl ). Shattering Ionic compounds are generally hard, but brittle. What we need is a second Na atom to donate a second electron to the O atom: Individual atoms can gain or lose electrons. The O atom still does not have an octet of electrons. Compounds formed from positive and negative ions are called ionic compounds. This interaction typically occurs between a metal, which carries positive. \) and hypochlorite is ClO −.\nonumber \] Ionic compounds form when atoms or molecules attract others with opposite charges. The compound is neutral overall, but consists of positively charged ions called cations and negatively charged ions called anions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
